Welcome to our page!
Click on the navigation toolbar for other pages.
UNIT 2 — RADICALS AND OZONE
What are radicals?
Radicals are defined as uncharged species with one or more unpaired electrons. As such, they are highly unstable.
How are radicals formed?
The formation of radicals may involve the breaking of covalent bonds by homolysis, a process that requires significant amounts of energy.
In our daily lives, there are several ways radicals can be formed:
Why is this important?
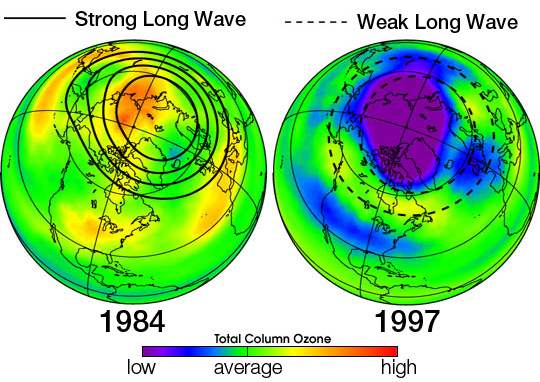
In this topic, our group investigates the effects of radicals on the ozone layer.
Radicals produced by various processes can lead to the destruction of the ozone layer, which in turn can lead to negative environmental consequences on Earth and for humans. Specifically, our group will focus more on the effects of chlorine radicals (produced by chlorofluorocarbons (CFCs) and hydrochlorofluorocarbons (HCFCs)) on the ozone layer – the formation of chlorine radicals, their effect on the ozone layer and the implications of ozone destruction.
In the recent few years, our ozone has been depleting rapidly. Therefore, there is a need to address this issue (and the chemistry behind it) and discuss its implications on our society, clarifying any misconceptions people may have regarding the depletion of ozone layer.
References
Radical (Chemistry). (n.d.). In Wikipedia. Retrieved March 20, 2015, from http://en.wikipedia.org/wiki/Radical_%28chemistry%29#Formation