The Context Of Polyethylene Today
Over 60 million tonnes of polyethylene manufactured each year making it the world’s most important thermoplastic. Thermoplastics have some specific properties, for example, they have no chemical crosslink, can be easily moulded when heated which is a reversible process and can return to their original hardness/strength when cooled. Therefore, polyethylene can be manufactured in many processed like extrusion, injection moulding, casting, etc. and its uses include film, packaging and containers, from bottles to buckets.
LDPE has a high degree of branching, meaning the chains don’t pack into the crystal structure as well. It has, therefore, weaker intermolecular forces as the instantaneous induced dipole – dipole attraction is less. This results in a lower tensile strength but increased ductility. The high degree of branching with long chains gives molten LDPE unique and desirable flow properties. LDPE is used for rigid containers, plastic film applications such as plastic bags and film wrap and for electrical insulation.
HDPE has a low degree of branching and thus has increased stiffness and decreased tensile strength. Compare to LDPE, HDPE is harder, tougher and has higher melting point. HDPE is used in products and packaging such as milk jugs, detergent bottles, butter tubs, garbage containers and water pipes. One third of all toys are manufactured from HDPE.
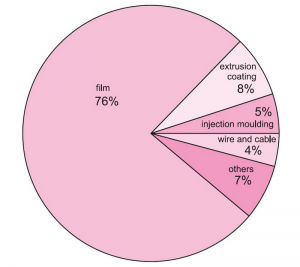
Products Made From Plolyethelene
Environmental Issues
Although ethylene can be produced from renewables, polyethylene is mainly made from petroleum or natural gas.
One of the main problems of polyethylene is that without special treatment it is not readily biodegradable, and thus accumulates.
Studies have shown that environmental exposure to plastic-related chemical compounds has adverse effects on human health. Due to the complexity of the substances that is released into the environment and the potential interaction of these substances, many questions exist on the safety of plastics for humans and the environment.
Alternatives To Polyethylene
One of the solutions is to biodegrade plastic by various ways. For example, Pseudomonas fluorescens, with the help of Sphingomonas, which was discovered in 2008 by a 16-year-old Canadian boy, Daniel Burd, who won the Canada-Wide Science Fair in Ottawa. It can degrade over 40% of the weight of plastic bags in less than three months. Another example is the prototype of a machine which creates oil from polyethylene using a small, self-contained vapor distillation process, which was discovered in 2010.
Another suggestion is bio-derived polyethylene. In other words, polyethylene can be produced from sugar cane. It can also be made from other feedstocks, including wheat grain and sugar beet. Bio-polyethylene may be promising, but it is not a perfect solution since bio-polyethylene is more expensive to produce, and many uses plant such as corn, resulting in competition with food supply. These developments using renewable resources rather than fossil fuel are a good start in our efforts towards environmental preservation.
Reference:
1. http://en.wikipedia.org/wiki/Polyethylene
2. http://en.wikipedia.org/wiki/Renewable_Polyethylene
3. http://www.essentialchemicalindustry.org/polymers/polyethene.html
4. http://journalistsresource.org/studies/environment/pollution-environment/plastics-environmental-health-literature-review