Welcome to the colourful world of…
TRANSITION METALS

What are Transition Metals?
- Make up the d-block and f-block (inner transition elements) of the periodic table.
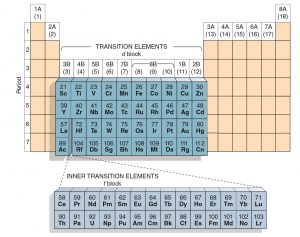
- Form cations with multiple oxidation states eg. Manganese can exhibit +2, +4, +7 oxidation states, just to name a few.
- These cations are often the center of many complex ions, known as Coordination Complexes. These species are responsible for the color of gems such as rubies (Cr3+) and sapphires (Fe2+, Fe3+ and Ti4+).
A More Rigorous Definition
A transition metal is one which forms one or more stable ions which have incompletely filled d orbitals.
Take note that while all transition metals are d-block elements, NOT all d-block elements are transition metals. (By the above definition)
For example,
Zn2+: [Ar]3d10 is NOT a cation of a transition element because the 3d orbitals of Zn are completely filled.
Fe2+: [Ar]3d6 is a cation of a transition element because the 3d orbitals of Fe are incompletely filled.
Coordination Complexes
- Formed with several molecules or ions (called ligands) surrounding a central metal ion, held by dative covalent bonds (called coordinate bonds)
- If the coordination complex carries a net charge, the complex is called a Complex Ion.
- Ligands act as Lewis bases and have at least one lone pair of electrons available for dative bonding.
- Two types of ligands:
- Monodentate: Have only one atom capable of binding to a central metal atom or ion eg. H2O, CN–, OH–
- Multidentate: Possess lone pairs on different atoms that are separate enough such that both can reach the metal eg. Ethylenediamine
- A complex ion containing a multidentate ligand is called a chelate.